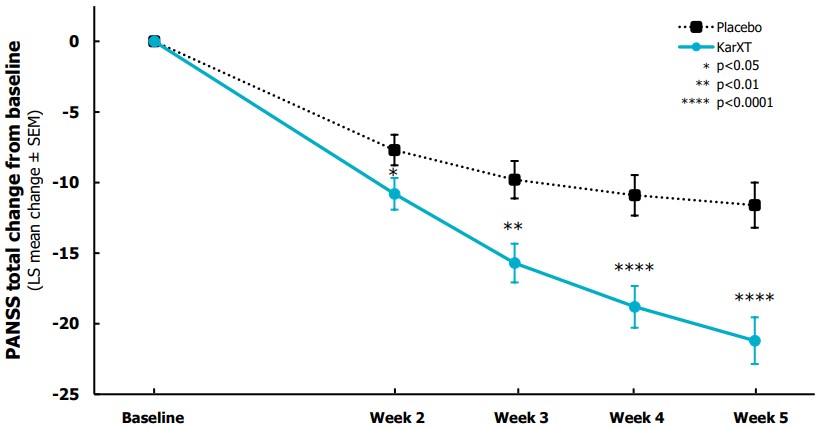
Otsuka and Lundbeck Announce U.S. Food and Drug Administration (FDA) Approval of Supplemental New Drug Application (sNDA) for REXULTI® (brexpiprazole) for the Treatment of Agitation Associated with Dementia Due to Alzheimer's Disease
Por um escritor misterioso
Last updated 03 janeiro 2025

Inline XBRL Viewer

News Releases Otsuka Pharmaceutical Co., Ltd.

FDA - Denmark

Piyush Dham on LinkedIn: The caring company: At Otsuka America

Dementia - Drug Discovery World (DDW)

Mischa Agster on LinkedIn: Otsuka Announces New FDA Approval
Clinical Catch-Up: Alzheimer's, MS and Cancer Headline Trial News

Allison Rosenthal on LinkedIn: InTOW Accepting Applications for
H. Lundbeck A/S and Otsuka Pharmaceutical, Co. Ltd. Announce U.S.

Otsuka and Lundbeck Announce U.S. Food and Drug Administration

Allison Rosenthal on LinkedIn: #worldmentalhealthday

Allison Rosenthal على LinkedIn: National Family Caregivers Month

Otsuka, Lundbeck's sNDA for Rexulti received US FDA approval

Dementia - Drug Discovery World (DDW)
Recomendado para você
-
 FDA's Fast-Track for Rexulti Raises Concerns03 janeiro 2025
FDA's Fast-Track for Rexulti Raises Concerns03 janeiro 2025 -
 Otsuka America Pharmaceutical 59148003613 - McKesson Medical-Surgical03 janeiro 2025
Otsuka America Pharmaceutical 59148003613 - McKesson Medical-Surgical03 janeiro 2025 -
 Rexulti (Brexpiprazole) - PSYCH-MENTAL HEALTH HUB03 janeiro 2025
Rexulti (Brexpiprazole) - PSYCH-MENTAL HEALTH HUB03 janeiro 2025 -
 Rexulti side effects and how to avoid them - NiceRx03 janeiro 2025
Rexulti side effects and how to avoid them - NiceRx03 janeiro 2025 -
Brexpiprazole (Rexulti): Uses, Side Effects, Warnings & More - GoodRx03 janeiro 2025
-
 Rexulti Works for Agitation in Alzheimer's, Despite Risks, Says FDA Staff03 janeiro 2025
Rexulti Works for Agitation in Alzheimer's, Despite Risks, Says FDA Staff03 janeiro 2025 -
 Precio Rexulti 1 mg 10 con tabletas03 janeiro 2025
Precio Rexulti 1 mg 10 con tabletas03 janeiro 2025 -
 Rexulti Advertisement Poster for Sale by BLTC03 janeiro 2025
Rexulti Advertisement Poster for Sale by BLTC03 janeiro 2025 -
 Rexulti Alternatives We Offer Natural, Evidence-Based Options03 janeiro 2025
Rexulti Alternatives We Offer Natural, Evidence-Based Options03 janeiro 2025 -
 REXULTI 2 MG Oral Tablet03 janeiro 2025
REXULTI 2 MG Oral Tablet03 janeiro 2025
você pode gostar
-
 Melhores animes para assistir na Netflix03 janeiro 2025
Melhores animes para assistir na Netflix03 janeiro 2025 -
 How To Play As Rosa In Bayonetta 203 janeiro 2025
How To Play As Rosa In Bayonetta 203 janeiro 2025 -
 12-Year-Old Master Tani Adewumi Granted Asylum in U.S.03 janeiro 2025
12-Year-Old Master Tani Adewumi Granted Asylum in U.S.03 janeiro 2025 -
 NEW* ALL WORKING UPDATE 2 CODES FOR ANIME WARRIORS SIMULATOR 203 janeiro 2025
NEW* ALL WORKING UPDATE 2 CODES FOR ANIME WARRIORS SIMULATOR 203 janeiro 2025 -
 Jogos de Meninas03 janeiro 2025
Jogos de Meninas03 janeiro 2025 -
 Bad Time Simulator (Sans Fight)-Undertale Game complete Detail03 janeiro 2025
Bad Time Simulator (Sans Fight)-Undertale Game complete Detail03 janeiro 2025 -
 Esporte Agil Galeria Fotos03 janeiro 2025
Esporte Agil Galeria Fotos03 janeiro 2025 -
 Game of Thrones GIFs on GIPHY - Be Animated03 janeiro 2025
Game of Thrones GIFs on GIPHY - Be Animated03 janeiro 2025 -
 Mortal Kombat Mobile: Gerador Ilimitado de Moedas e Almas 202303 janeiro 2025
Mortal Kombat Mobile: Gerador Ilimitado de Moedas e Almas 202303 janeiro 2025 -
 Sono Bisque Doll wa Koi wo Suru Cosplay Manga gets TV Anime!!03 janeiro 2025
Sono Bisque Doll wa Koi wo Suru Cosplay Manga gets TV Anime!!03 janeiro 2025